Iso 13485 compliance update#

GD211 Training, Module 2 - Information about the manufacturer.GD211 Training, Module 1 - Introduction.
Iso 13485 compliance portable#
The Training Modules are available in Microsoft PowerPoint and Portable Document Format as an alternate format to the Study Guide. The following Training Modules summarize the contents of the Study Guide GD211: Guidance on the content of quality management system audit reports and are available as a convenience to the user. Study Guide GD211: Guidance on the content of quality management system audit reports.Form F202 Submission of a New or Modified Quality Management System Certificate.
Iso 13485 compliance registration#
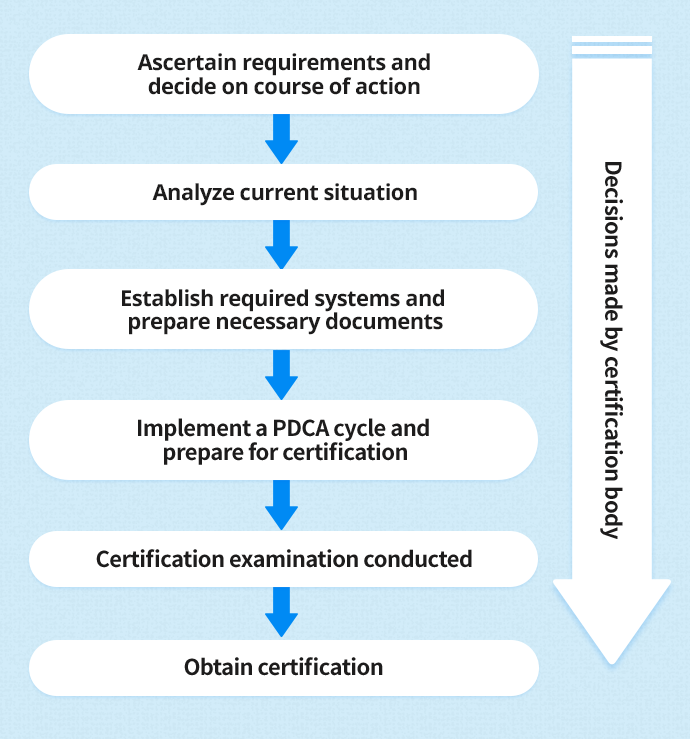
There are no regulatory quality system requirements for Class I medical devices. The Medical Devices Regulations require class II, III and IV medical devices to be manufactured (class II) or designed and manufactured (class III & IV) under CAN/CSA ISO 13485:2003. The Medical Devices Regulations do not require importers or distributors of medical devices to have a registered quality system. Health Canada will only accept quality system certificates that have been issued by special third party auditing organizations called Canadian Medical Devices Conformity Assessment System (CMDCAS) recognized registrars. Health Canada requires medical device manufacturers to use a quality system certificate as evidence of compliance to the appropriate regulatory quality system requirement.
